This article has mentions of products from one or more companies, and I may receive compensation if you purchase those products following reading my recommendations.
When it comes to choosing the perfect diamond, color is a crucial factor that can greatly impact the gem's overall appeal and value. As a diamond expert, I've put together this comprehensive guide to help you understand the intricacies of diamond color, from the widely recognized grading system to the rarity and value of fancy-colored diamonds. By the end of this article, you will have a solid understanding of diamond color, empowering you to make well-informed decisions when selecting or investing in these precious stones. So, let's dive into the fascinating world of diamond color and discover what makes each hue unique and valuable.
The Diamond Color Grading System
The GIA Color Scale
The Gemological Institute of America (GIA) developed a widely accepted color grading system for diamonds, which has become the industry standard. This system provides a consistent and objective framework to evaluate and communicate diamond color, facilitating the comparison of diamonds and ensuring an accurate representation of their quality.
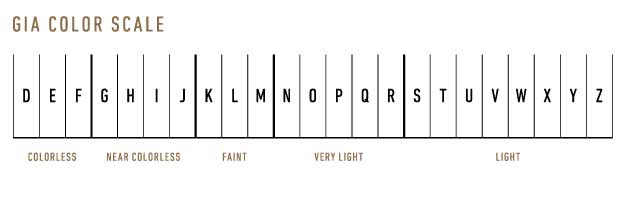
Grading process: D-Z color scale:
The GIA color scale is an alphabetical scale that ranges from D (colorless) to Z (light yellow or brown). Diamonds are compared to master stones under controlled lighting conditions and a neutral background to accurately determine their color grade. The color distinctions are often subtle, and the grading process requires highly trained gemologists and specialized equipment.
Importance of the GIA color scale in the diamond industry:
The GIA color scale is essential for maintaining trust and transparency within the diamond industry. It provides a universal language for grading and discussing diamond color, helping buyers and sellers make informed decisions when purchasing, selling, or valuing diamonds.
The IGI Color Scale
The International Gemological Institute (IGI) is another well-respected organization in the diamond industry that also provides diamond color grading services. Similar to the GIA, the IGI uses a color scale that ranges from D (colorless) to Z (light yellow or brown). While the GIA is often considered the gold standard for diamond grading, the IGI is a credible alternative that offers reliable and consistent grading.
Grading process: D-Z color scale:
The IGI color grading process closely resembles that of the GIA, with diamonds being assessed in a controlled environment using standardized lighting conditions and a neutral background. IGI-trained gemologists compare diamonds to master stones to determine their color grade accurately.
Importance of the IGI color scale in the diamond industry:
The IGI color scale plays a significant role in the diamond industry, providing an alternative to the GIA for diamond grading. It helps maintain consistency and transparency within the industry, giving buyers and sellers a trusted source for diamond grading and ensuring a fair evaluation of diamond color.
Understanding Diamond Color Grades

Colorless diamonds (D-F):
The highest color grades, D, E, and F, are considered colorless diamonds. These diamonds exhibit no trace of color and are the most valuable due to their rarity. They allow the maximum amount of light to pass through, resulting in exceptional brilliance and fire.
Near colorless diamonds (G-J):
Near colorless diamonds, graded G, H, I, and J, exhibit only slight traces of color. To the naked eye, these diamonds appear nearly as colorless as D-F diamonds, making them a popular and more affordable option for many buyers. The subtle color difference is often undetectable in most settings.
Faintly colored diamonds (K-M):
Diamonds graded K, L, and M display a faint yellow or brown coloration. While the color may be noticeable, especially when compared to colorless diamonds, these stones can still be desirable in specific settings or styles that complement their hue.
Very light-colored diamonds (N-R):
Diamonds in the N-R color range exhibit a very light yellow or brown color. These diamonds are less expensive and less sought after, as their color can detract from their brilliance.
Light-colored diamonds (S-Z):
Diamonds with a color grade of S-Z display a light yellow or brown color. These diamonds are even less valuable and less popular than those in the N-R range. However, they may be suitable for buyers seeking a unique appearance or a lower-priced option.
Factors Affecting Diamond Color
Chemical impurities
1. Nitrogen: Yellow and brown diamonds
The presence of nitrogen atoms in a diamond's crystal lattice can cause yellow and brown hues. Nitrogen atoms absorb blue light, allowing yellow light to be transmitted and reflected, resulting in a yellow appearance. The concentration of nitrogen and its distribution within the diamond determines the intensity of the color.
2. Boron: Blue diamonds
Blue diamonds are the result of boron impurities in the diamond's crystal lattice. When boron atoms replace carbon atoms in the lattice, they absorb red, yellow, and green light, causing the diamond to appear blue. The rarity of boron impurities in diamonds makes blue diamonds highly sought after and valuable.
Crystal Structure Defects
1. Plastic deformation: Pink, red, and brown diamonds
Pink, red, and brown diamonds owe their color to plastic deformation in the crystal lattice. This deformation causes selective absorption of light, resulting in unique colors. The exact cause of the deformation is still debated among scientists, but it is believed that extreme pressure and temperature during diamond formation play a role.
2. Radiation exposure: Green diamonds
Green diamonds are formed when naturally occurring radiation in the Earth's crust alters the diamond's crystal lattice. This alteration causes the diamond to absorb red and yellow light, making it appear green. The depth and intensity of the green color depend on the level and duration of radiation exposure.
Natural vs. treated diamonds
1. Natural coloration
Natural colored diamonds are formed due to the presence of chemical impurities or crystal lattice defects, as mentioned above. These diamonds are highly prized for their rarity and unique colors, which are created over millions of years under specific geological conditions.
2. Color-enhancing treatments
Some diamonds undergo color-enhancing treatments to improve or modify their color. While treated diamonds may be more affordable than their natural counterparts, it is important to be aware of these treatments when purchasing diamonds.
a. High-pressure, high-temperature (HPHT) treatment
HPHT treatment involves subjecting a diamond to high pressure and high temperature to change its color. This treatment can remove or lessen yellow or brown coloration in diamonds, making them appear more colorless. It can also create fancy-colored diamonds by modifying the existing color. This is also the process used to create lab-grown diamonds.
b. Irradiation and annealing
Irradiation and annealing treatments involve exposing a diamond to radiation, followed by controlled heating to change its color. These treatments can create a wide range of fancy-colored diamonds, including blues, greens, yellows, and browns.
c. Coating
Coating treatments involve applying a thin layer of material to the diamond's surface to alter its color. While this method can produce visually appealing results, it is less durable and can be removed with wear or during repairs.
Fancy Colored Diamonds
Overview of fancy colored diamonds

Fancy-colored diamonds are diamonds that display colors beyond the D-Z scale used for grading colorless to light yellow or brown diamonds. These diamonds exhibit vibrant and rare hues, making them highly sought after and valuable. Fancy-colored diamonds can occur naturally or be created through color-enhancing treatments.
Rarity and value of fancy colored diamonds
Natural fancy-colored diamonds are rare and often command higher prices compared to colorless diamonds of similar size and quality. The rarity of certain colors, combined with their intensity and evenness, can significantly impact the value of a fancy-colored diamond. Some of the rarest and most valuable fancy colors include red, blue, and pink.
Popular fancy colors
1. Yellow: Fancy yellow diamonds, also known as canary diamonds, are relatively common in the world of fancy-colored diamonds. Their color ranges from light to deep yellow and can be influenced by the presence of nitrogen impurities.
2. Pink: Pink diamonds are incredibly rare and highly sought after. Their color is believed to result from plastic deformation during the diamond's formation. The Argyle mine in Australia is a significant source of pink diamonds.
3. Blue: Blue diamonds are extremely rare and valuable. Their color is attributed to the presence of boron impurities in the diamond's crystal lattice. The famous Hope Diamond is an example of a blue diamond.
4. Green: Green diamonds are the result of natural radiation exposure. Their unique color is caused by the absorption of red and yellow light. Natural green diamonds are very rare, as the green color is often limited to the diamond's surface.
5. Orange: Orange diamonds are rare and desirable. Their color is attributed to nitrogen impurities and crystal lattice deformation. The intensity of the color can range from light to deep orange.
6. Red: Red diamonds are among the rarest and most valuable fancy-colored diamonds. Their color is believed to result from plastic deformation, similar to pink diamonds.
7. Purple: Purple diamonds are rare and often exhibit a color range from light lavender to deep purple. Their color is thought to be caused by crystal lattice deformation combined with the presence of hydrogen impurities.
8. Gray: Gray diamonds are less common and can exhibit a range of hues, from light to dark gray. Their color is often due to the presence of hydrogen impurities or inclusions of other minerals.
9. Brown (chocolate, cognac, champagne): Brown diamonds are relatively more common and can be found in shades such as chocolate, cognac, and champagne. Their color is often caused by plastic deformation and/or the presence of nitrogen impurities.
10. Black: Black diamonds are opaque and absorb all light, resulting in their black appearance. Their color is typically due to the presence of numerous inclusions or graphitic material.
Grading fancy colored diamonds
1. Hue, tone, and saturation: Unlike the D-Z color scale, fancy colored diamonds are graded based on hue, tone, and saturation. Hue refers to the dominant color of the diamond, tone refers to the lightness or darkness of the color, and saturation refers to the color's intensity or purity. The combination of these factors determines the overall color grade and value of a fancy-colored diamond.
2. The role of the GIA in grading fancy-colored diamonds: The GIA also grades fancy-colored diamonds using a specialized system. They evaluate the diamond's hue, tone, and saturation to assign a grade that accurately represents the diamond's color. This grading process helps ensure consistency and transparency in the fancy-colored diamond market.
The Impact of Diamond Color on Value and Aesthetics
The relationship between color and price
The color of a diamond has a significant impact on its price. In general, colorless diamonds (D-F) command the highest prices due to their rarity and exceptional brilliance. As diamonds move down the color scale, their price typically decreases. However, fancy-colored diamonds can be an exception, as their rarity and unique hues can command higher prices than colorless diamonds.
Choosing the right color for different settings and styles
When selecting a diamond, it's essential to consider how its color will interact with the setting and style of the jewelry. For example, near-colorless diamonds (G-J) can look virtually colorless when set in yellow gold, while colorless diamonds may appear more striking in white gold or platinum settings. Additionally, certain fancy-colored diamonds can look stunning when paired with complementary gemstones or metal colors that enhance their hues.
Balancing diamond color with other quality factors (cut, clarity, and carat)
While color is an important aspect of a diamond's overall appeal and value, it's essential to consider the other quality factors as well. The cut, clarity, and carat weight of a diamond can greatly impact its appearance and price. A well-cut diamond can enhance its color and overall beauty, while high clarity ensures the diamond is free of visible inclusions that can detract from its brilliance. Balancing these factors with color is crucial when selecting the perfect diamond for your needs and budget.
Tips for Buying Diamonds Based on Color
Determine your personal preferences and priorities
Before purchasing a diamond, take the time to consider your personal preferences and priorities. Some buyers may prioritize a higher color grade, while others may prefer to focus on other quality factors, such as cut, clarity, or carat weight. Understanding what is most important to you will help guide your decision-making process.
Consider the setting and metal color
As mentioned earlier, the setting and metal color can significantly influence the appearance of a diamond. Near colorless diamonds (G-J) may appear colorless in yellow or rose gold settings, while colorless diamonds (D-F) may be more striking in white gold or platinum settings. Be sure to consider how the diamond color will interact with the setting and metal color when making your selection.
View diamonds in different lighting conditions
Diamonds can appear differently under various lighting conditions. It's essential to view diamonds under different types of lighting, including natural daylight, fluorescent lighting, and incandescent lighting, to get a true sense of their color. This will help you make a more informed decision when choosing a diamond based on color.
Compare diamonds side by side
When possible, compare diamonds side by side to see how their colors differ. This can help you better understand the subtle differences between color grades and make a more informed decision about which diamond best suits your preferences and budget.
Work with a reputable jeweler or gemologist
Request a grading report
When purchasing a diamond, always request a grading report from a reputable organization like the GIA or IGI. This report will provide an objective evaluation of the diamond's color, as well as its other quality factors. This information will help ensure that you are making an informed decision when selecting a diamond based on color.
Be aware of color-enhancing treatments
As mentioned earlier, some diamonds undergo color-enhancing treatments to improve or modify their color. Be sure to ask about any treatments the diamond may have undergone and how they may impact the diamond's value and durability. Working with a reputable jeweler or gemologist can help ensure that you are aware of any treatments and can make an informed decision when purchasing a diamond based on color.
Conclusion
The color of a diamond has a significant impact on its overall appearance and value. When selecting the right diamond based on color, it's important to understand your personal preferences and priorities, consider the setting and metal color, view diamonds in different lighting conditions, compare diamonds side by side, work with a reputable jeweler or gemologist, request a grading report, light performance images, and be aware of any potential treatments. By following these tips, you can ensure that you are making an informed decision when selecting the perfect diamond for your needs and budget.
